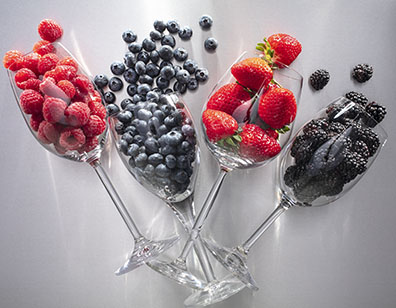Bottling & Aging Wine
Fill It Up
With all the preparatory work — rinsing, sanitizing and more rinsing — bottling can become overwhelming as one’s production increases. That can quickly turn into many hours of bottling using a single siphon and racking cane.
Stem-and-Valve bottle filler
The most basic bottling apparatus is the stem-and-valve bottle filler, also known as a bottling wand, which is ideal for 5-gallon (19-L) batches. It typically retails for approximately $5 or less. It can fill 30 standard 750-mL bottles in 20 minutes. These fillers consist of a short plastic tube with a stem attachment. The stem has either a simple or spring-activated valve to control the flow of wine, and uses gravity to displace wine from a carboy to bottles. To fill a bottle, you simply prime the filler, insert it in an empty bottle, and apply pressure by pressing the stem against the bottom of the bottle. Release the pressure when the wine level reaches the top of the bottle. When you remove the filler, the wine level will fall back down to the desired level for inserting standard corks.
The stem-and-valve bottle filler works well for standard flat-bottom bottles but it is hard to operate when the bottle has a deep punt — an indentation in the bottom of bottles. The stem will slide down the punt, and therefore the valve never opens.
Semiautomatic fillers
There are many types of semiautomatic bottle fillers available, but cost can become an issue. If you’re interested, most shop owners should be able to assist you with purchase information.
Corking
After you have your wine in the bottle, it is important to get it sealed up. The traditional method is to insert a type of cork into the neck of your bottle (see chart for a list of corks and prices).
Aging Potential: Understanding the lifespan of your wine
by Daniel Pambianchi
For many wine lovers, the subject of a wine’s aging potential can be intimidating or seem like artful science that is best left to the wine gurus of the world. Some are often misguided by false myths that all wines can age or that homemade wines cannot age.
We can use our knowledge of the wine’s chemistry and organoleptic attributes (color, odor, taste and feel) to evaluate how long a wine can last and when it will be best to drink. With experience, we’ll be able to estimate the life of a wine by relying solely on its organoleptic attributes — like the pros who do not have access to analytical data when tasting wine.
Wine is first created in the vineyard as the soil, climate and viticultural practices (e.g. crop thinning, timing of harvest, etc.) dictate the quality and chemistry of the fruit. Only the best fruit coupled with sound winemaking methods will yield age-worthy wines. In general, reds will age better than whites; however, the vast majority of wine produced globally is meant for drinking as soon as it is marketed or within a year or two. Only a small percentage of wine, about 3–5%, is meant for extended cellaring. Homemade wine can be made to last many years.
The wine’s chemistry — namely, color, pH, acidity, tannins and alcohol content — plays an important role. Each indicator impacts aging potential, however, they must be considered holistically for a more accurate assessment. If any one indicator is abnormally low or high for a specific style of wine, the wine will be unbalanced and may not age gracefully.
Understanding the indicators will not provide an exact assessment in years, but will give you some clues as to when the wine should be drunk. As you acquire tasting experience and follow a wine’s evolution over time, your estimates will greatly improve.
Acidity, pH and Color
A wine’s pH and color are the most important indicators when assessing aging potential. Lisa Van de Water, of the Wine Lab at Napa, best summarized the effect of pH on wines, as reported by Donald E. Gauntner in the American Wine Society Journal (Winter Issue, 1997).
“At lower pH, red wines are redder, fresher, fruitier, younger tasting for their age, slower to age, slower to mature, less complex, less full-bodied, much slower to spoil, and easier to maintain free of spoilage in the cellar because the SO2 is more active.”
“Higher pH wines, if they are red, are less red (sometimes brown, sometimes purple), less fresh, less fruity, more complex, more full-bodied, faster to age, faster to mature, easier to spoil, and more difficult to manage in the cellar with SO2.”
Wine typically has a pH between 3.2 and 3.6 with whites at the lower end and reds at the higher. Below this range, the wine’s high acidity may make it unbalanced, and above this range it will be unstable and dull. In whites, the color will darken to a gold color and turn to a brownish color if aged too long. In reds, the color will lighten and take on an orangey color, followed by a brownish color, most noticeable at the rim. If color and pH suggest that the wine is in its prime, reassess the wine at least once a year. Keep records of each wine’s evolution to understand its aging dynamics.
Acidity has an inverse relationship to pH: A low-pH wine will have a higher acidity and a high-pH wine will have a lower acidity. As such, the refreshing acidity in wine is a good indicator of its aging potential. A flabby or flat wine lacks acidity essential for aging and should be drunk as soon as possible.
Tannins
In red wines, tannins are responsible for body and aging potential, and interact with color pigments to provide color stability. They are essential in making age-worthy wines. Tannins are anti-oxidants and, therefore, mouth-puckering, tannin-rich wines will live longer than low-tannin wines. A wine described as “full-bodied” implies that the tannin content is high while one described as “not approachable in its youth” implies that the tannins are too harsh and require aging to soften.
White wines are very low in tannin concentration and are therefore not meant for aging. The best white wines aged in oak barrels can live longer however, because of the higher tannin content. Similarly, wines (e.g. Beaujolais Nouveau) made by carbonic maceration — a technique used to trigger fermentation within whole berries to extract maximum fruit flavors but with no tannin extraction — are not meant for aging being low in tannin content and malic acid.
Alcohol
High alcohol levels in wine is often (mistakenly) interpreted as “full-bodied.” A low-alcohol wine can be full-bodied while a high-alcohol wine can lack body. However, alcohol acts as a preservative, and therefore, higher-alcohol wine will generally outlive lower-alcohol wine.
For example, a tannin-rich, 20%-alcohol port wine can age for decades whereas a fruity, 7%-alcohol Muscat wine should be drunk much earlier. In contrast, a syrupy sweet, 10%-alcohol icewine can be aged ten years or more owing to its higher acidity.
Aroma and Bouquet
A wine’s aroma and bouquet can reveal important clues about a wine’s aging potential. Wines are often described as “closed in,” and therefore requiring cellaring to achieve their full potential. Let’s first understand the difference between aroma and bouquet. Émile Peynaud — an authoritative research enologist and teacher of modern winemaking — best describes these terms in The Taste of Wine: The Art and Science of Wine Appreciation (Michael Schuster, tr., Macdonald & Co, 1987). Aroma is “the sum of (odor) elements in young wines” and bouquet is “the smells acquired through (aging), which develop gradually over the course of time.”
“Defined in this way young wines owe their charm more to their aroma than their bouquet, while the appeal of wines with several years of bottle age will be due entirely to their bouquet.”
Young wines will tend to exhibit aromas acquired from the grape varieties used and vinification methods, such as yeast selection. They may lack complexity and therefore seem “closed in” as the aromas have not had a chance to evolve and transform into more flavors as a result of oxidation and complex chemical reactions. Once transformed from aging, the bouquet can exhibit multiple flavors — the more flavors, the more complex.
How can one tell if a wine is past its prime judging from its bouquet? Assuming that the wine has not acquired any faults from poor winemaking or poor cellaring, the wine might well be past its prime if the bouquet seems one-dimensional or has off-odors. The wine is said to be fatigued, having lost much of its fruity aromas and subtle bouquet. Off-odors may include a vinegar smell or a pronounced nutty flavor (usually accompanied by a brownish color).
Free SO2
Free SO2 is not a factor when evaluating aging potential; however, it is used as a preservative to ward off microbial spoilage and premature oxidation. Although it helps the wine, sulfite addition should not be mistaken as a means to extend the life of wine. The recommended nominal free SO2 level is about 10 times the decimal value of the wine’s pH. For example, a red wine with a pH of 3.3 requires 30 mg/L; add an extra 10 mg/L for whites.
Cellaring
Remember the three cardinal rules of proper wine cellaring if you intend to age wine.
1. Maintain a constant cool cellar temperature in the 54–59 °F (12–15 °C) range.
2. Maintain relative humidity in the 65–75% range.
3. Protect wine from vibrations, light and odors.
Bad oxygen
by Daniel Pambianchi
We often read that air (and especially the oxygen it contains) is a wine’s worst enemy. Oxidized wine becomes devoid of subtle and fruity aromas that make it seem tired, as if it is well beyond its apogee (even though it is not). Having lost its finesse and intensity, the wine will also take on a brownish color. As oxidation progresses, the wine will take on a deeper brown color with a sherry-like smell and eventually spoil if untreated. When spoiled, the wine will develop a heavy, white film on the surface and possibly a bluish green mold. This primer will help you understand the harmful effects of oxidation and guide you in preventing, or at least reducing the probability of oxidation.
Understanding Oxidation
Simply stated, oxidation is the result of wine being exposed to air during winemaking, at any time from grape or juice handling to bottling and from an inadequate sulfiting regimen. The more technical explanation is that free oxygen in air causes an enzymatic reaction with oxidases — the oxidizable enzymes, such as tyrosinase (polyphenoloxidase) — found in grape juice resulting in oxidation of aromatic and pigmentation compounds.
Fermentation and vinification compounds found in wines — such as alcohol, acidity, tannins and sulfur dioxide by-products — offer better oxidation protection than musts. Oenologist Emile Peynaud supports this theory in his book “Knowing and Making Wine” (John Wiley & Sons, 1984) by stating that “musts consume on the average two milligrams of oxygen per liter per minute whereas wines take 24 hours to consume the same amount.”
The susceptibility of must to oxidation underlines the extra care required during grape and juice handling. In general, red wines are less prone to oxidation effects because of their higher concentration of phenolic compounds, which inherently ward off hungry oxygen molecules.
The enzymatic reaction also accelerates as temperature increases, which increases oxidative effects and therefore requiring the wine to be stored at a cool temperature, for example around 55 °F (13 °C). Additionally, as pH increases must and wine become more susceptible to oxidative effects. Therefore, low pH wine is better protected and will tend to age better and longer and exhibit more aromas and a livelier color. And remember, the effectiveness of free SO2 from sulfite additions for protecting wine against oxidation (and spoilage organisms) also decreases as pH increases.
10 Ways to Ward Off Oxidation
There are two general recommendations to follow diligently during winemaking: 1) minimize the must’s and wine’s exposure to air, and 2) sulfite judiciously. Following are ten specific reccomendations to avoid oxidation.
1. Transfer Whites Quickly:White musts are most prone to the effects of oxidation and should therefore be immediately transferred to carboys following crushing and pressing operations. White wine maceration is not recommended; otherwise, the wine will turn a brownish color. Be sure to remove berries and grape bunches affected by mold or rot.
2. Adjust the pH: The higher the pH, the more prone wine will be to oxidative effects. If your wine’s pH falls in the ideal range of 3.1–3.6, don’t play with it. To decrease the pH, add tartaric acid crystals before the start of fermentation at the rate of 1 g/L (0.13 oz./gallon) of juice for every 0.1 unit of pH.
3. Use Sulfite: Sulfur dioxide (SO2) is used in winemaking as a preventative against microbial spoilage, but also to reduce the effects of oxidation. And as pH increases, the effectiveness of free SO2 decreases. My rule of thumb is to maintain a nominal free SO2 level in a wine throughout its life until bottling at a value equivalent to 10 times the fraction value of a red wine’s pH, and add 10 for white wine. For example, for a red wine with a pH of 3.4, maintain a free SO2 level of 40 mg/L (ppm). A white wine with the same pH would require 50 mg/L. Use the sulfite calculator at www.winemakermag.com to determine how much sulfite you need to add for the desired free SO2 level.
4. Top Up: One advice that cannot be repeated often enough is to always, always top up your wood barrels or other containers when storing or aging wine. Following the alcoholic fermentation, fill your carboys to leave no more than 3⁄4 inch (1.9 cm) of ullage — the headspace between the surface of the wine and the bung. Then secure the carboy opening with a fermentation lock properly filled half way with water. Most oxidation problems related to inadequate top up involve barrel aging. Wine breathes through the wood and stave/head joints (i.e. all joints between any wood member) of the barrel. As wine evaporates, it is replaced with air. Check the wine level in all barrels at least twice a month and top up to the bung hole using a reserve wine of similar quality.
5. Rack by Gravity: It is often recommended to rack red wine “by splashing.” During racking, wine is allowed to splash at the bottom of the container and at the wine’s surface as the level rises in the container. This softens the wine and can increase the fruit characteristics. White wines should never be racked with splashing because these are more prone to oxidation. As much as possible, rack wine by gravity, as you first learned when you made that first batch of wine. Other mechanical means, such as pumps, which hasten oxidation should be avoided. Many commercial wineries pride themselves on having a gravity flow system, usually associated with higher quality wines because these have not been subjected to any mechanical processing.
6. Avoid Pumps: Unless you handle large volumes of wine where gravity flow is too time-consuming, the use of mechanical pumps should be kept to a minimum, such as in filtering. Mechanical pumps greatly speed up racking or wine transfer operations; however, they tend to dissolve some oxygen in the wine and therefore cause some oxidation. Always look for alternatives. For example, instead of (mechanical) pump over of wine during red wine maceration, punch down the cap. It is much gentler on the wine.
7. Use Closed Systems: When possible, use a closed system for winemaking operations to minimize wine exposure to air. For example, when filtering, use a vacuum pump to draw air out of the receiving carboy instead of using a normal pump.
8. Use Ascorbic Acid Diligently: Ascorbic acid (vitamin C) is an ingredient often used in home winemaking for its antioxidant properties. However, its use is not well understood. Ascorbic acid only has an extremely transitory antioxidant effect on wines. It fixes to dissolved oxygen in wine and quickly converts it to dehydroascorbic acid, a weak organic acid, within three or four days. Following this oxidation, ascorbic acid is exhausted and serves no further function. It is mainly used to scavenge oxygen in wine before bottling or other operations where the wine will be subjected to little or temporary aeration. Add ascorbic acid at a rate of 2–3 g/hL (approximately 1⁄2 tsp. per 25 gallons) of wine just before bottling. Be sure the wine has the minimum recommended level of free SO2 before adding ascorbic acid; otherwise, it might actually favor oxidation.
9. Store at a Cool: Temperature The ideal temperature is around 55 °F (13 °C). Don’t go lower than this temperature; otherwise, wine will take much too long to develop gracefully.
10. Inspect your Equipment: Winemaking equipment may develop defects over the year from wear and tear. Any defective equipment can easily spoil wine. Inspect all equipment regularly, and particularly any jointed or molded apparatus. For example, check around the valve at the bottom of your stainless steel tank, verify that bungs are not defective and that they form an airtight seal with their vessel and check for leaks in oak barrels.
Good Oxygen
by Daniel Pambianchi
Positive affects of oxidation? How can that be? Wine-makers know that oxygen negatively affects wine. A little oxygen, however, can actually be beneficial to your homemade wine.
Two common winemaking practices that have a positive influence are macroaeration and microoxygenation. Both are used in making some of the best reds and whites, evidenced by the rows and stacks of oak barrels in commercial wineries.
Macroaeration refers to the practice of aerating, or oxygenating, fermenting wine by vigorous racking to produce a softer, less astringent wine that exhibits more fruit character and improved color stability.
Microoxygenation refers to the controlled process of oak-barrel aging where wine is allowed to interact very slowly with air penetrating through the barrel. Apart from the flavors and tannins imparted by the wood, wines aged in oak barrels will exhibit more complexity than wines aged in stainless tanks or glass containers primarily because of microoxygenation.
Oxidation Reviewed
Oxidation is the result of wine being exposed to air during winemaking, at any time from grape or juice handling to bottling. Oxidation can also result from an inadequate sulfiting regimen. The more technical explanation is that free oxygen in air causes an enzymatic reaction with oxidases such as tyrosinase (poly-phenoloxidase) in grape juice resulting in the oxidation of the aromatic and pigmentation compounds.
Macroaeration
Macroaeration is only used in red winemaking as it involves vigorous splashing of wine during pump-over or racking operations. Macroaeration has a softening effect on the astringent tannins and enables the winemaker to produce a softer wine.
Wine aerated in this way is approachable much earlier (younger) and has well stabilized color. The higher phenolic content in reds affords them greater protection from negative oxidation effects compared to whites, which have considerably lower tannin content. Macroaeration therefore is never practiced and not recommended for white winemaking.
Macroaeration can also benefit red winemaking by providing yeast cells with oxygen and ensuring a healthy fermentation. Yeast require oxygen in order to thrive and to convert sugar into alcohol. If oxygen is not sufficiently available, yeasts can become inhibited and a stuck fermentation can result.
Another benefit is reduced hydrogen sulfide (H2S) production. A common cause of H2S in wines, detected as a rotten-egg smell, is vinification from grapes that have been over-treated with sulfur-based vineyard mildew and fungus inhibitors. Red wines made from grapes are more prone to H2S problems because the juice is allowed to macerate with the grape skins (therefore dissolving sulfite into the juice).
In white wines from grapes, it is not a problem because the juice is not macerated with the skins. Abundantly aerating the wine by racking it against the wall of the container will reduce the amount of potentially harmful H2S. If not treated early, H2S will react in the wine to form first into mercaptans followed by disulfides — both are foul-smelling compounds that cause wine to spoil. The presence of either compound is practically irreversible in home winemaking.
Macroaerating Wine
There are several ways to macro-aerate wine, each having a varying degree of effectiveness. These fall into two main categories: splashing wine and injecting oxygen into wine.
The most common method of splashing used by home winemakers is quite simple — just let the wine splash against the wall of a carboy during racking. Commercial wineries often use delestage fermentation, also known as the rack-and-return method. This method involves fermenting wine that is racked by gravity and flows into a vat, then returned to the tank with a pump. Refer to “Do the Delestage” in the June–July 2003 issue of WineMaker for more information on delestage fermentation.
Although these techniques are proven, some industry experts argue that the wine is still too protected by the layer of carbon dioxide gas that is forming over the wine volume during racking, and therefore the wine does not absorb sufficient air to benefit from macroaeration. It is winemakers of this opinion that turn to the method of injecting oxygen into the wine.
Commercial wineries connect a venturi attachment to their hose when returning wine to the top of the tank during the pump-over operation. The venturi attachment is a simple inverted T-connector that allows air
to be drawn into the wine stream when the wine is being pumped over. A check valve may also be used to prevent backflow.
A home winemaker can easily make a T-connector using polyethylene tubing and plastic barb adapters. The T-connector can then be inserted into the racking line by cutting the tube midway and connecting each end to the barb adapter. You will need to test with various lengths of tube on the leg side of the T until the “burping” stops during racking. Alternatively, if you use a pump with a valve on the suction side for pump-over, be sure to open this valve when working the pump.
And remember — do not aerate wine during malolactic fermentation (MLF). ML bacteria are usually very sensitive to oxygen and can become inhibited, which will commonly result in a stuck MLF.
Microoxygenation
A miniscule, controlled amount of oxygen during wine aging can be beneficial for wine to develop and show its full potential. It allows wine to develop and age gracefully while softening tannins, stabilizing phenols and increasing flavor complexity — a phenomenon known as microoxidation, or microoxygenation. This cannot happen in air-tight, inert containers such as stainless steel tanks or glass carboys.
Until recently, microoxygenation mainly occurred during barrel and bottle aging where wine breathes through the wood and head and stave joints (the tight space between the pieces of wood forming the heads and walls of a barrel). Wine can also breathe through the cork during the aging process.
New advances in winemaking technology however, now make it possible to microoxygenate wine in controlled fashion at any stage of the process, such as tank aging, for example, and replicate the benefits of barrel-aged wines, albeit in a much shorter time period.
Microoxygenating Wine
When barrel-aging wine, no special microoxygenation equipment or process is required; the barrel does all the work. The winemaker only has two tasks: The first is to check the wine level every three to four weeks to ensure that everything is developing as planned according to your desired taste. The second is to do is top up the barrel. This will avoid “bad” oxidation, offensive smells and unfavorable tastes in your wine.
For bottle-aging wine, natural or agglomerated corks are recommended. Some winemakers maintain that screw caps and synthetic corks provide too much of an airtight seal depriving the wine of the microoxygen that is necessary for it to evolve gracefully. The argument is that airtight seals inhibit microoxygenation and completely stifle the type of evolution required in well-aged wines. If a winemaker prefers to drink young wine, screw caps or synthetic cork closures are certainly a convenient method.
Keep Oxidation in Check
If done correctly, home winemakers can develop a means of letting oxygen work for them as opposed to against them and their wines. However, don’t forget about the negative effects of oxidation that discussed in the last article. Oxidized wine becomes devoid of fruity aromas and will spoil and need to be discarded if not treated. This can be discouraging for a beginning winemaker, so beware!