Using Winemaking Enzymes
Many winemakers shun the use of additives, including enzymes, to respect the wine’s “naturalness.” But juice is laden with natural enzymes, and once inoculated with yeast, fermenting wine is under the control of countless enzymes working hard to help convert odorless compounds into volatile, odiferous ones and create new ones, all of which will define the wine’s style and character.
Some enzymes can negatively affect quality and must therefore be kept in check; others are in short supply or non-existent and must therefore be added to craft a specific style of wine or simply to circumvent processing problems. Here, we will take a look at what enzymes are, how they work, and the types of exogenous enzymes (those you add) to help improve your winemaking and wines.

Enzymes
Enzymes are naturally occurring proteins with powerful catalytic activity, which are synthesized by biological cells in all organisms, and so they occur, for example, in plants, yeast and bacteria. A plethora of enzyme preparations are also available commercially to meet the needs of many industries, notably the food and beverage industry, where endogenous enzymes have insufficient activity potential due to limiting inhibitory factors (more on this later).
As catalysts, enzymes significantly increase the rate of reactions but do not undergo change. They accomplish this by reducing the amount of energy needed to enable those reactions, and the amount of energy required is a function of temperature, concentration of compounds involved in the reaction, and environmental factors such as pH.
Enzymes use what is known as a lock-and-key reaction model. They have an active site capable of accepting substrates. Once the substrates are locked into the active site, they react and the resulting products are freed, and the enzyme once again becomes available in its original molecular structure. For example, consider the case of a terpenol molecule, such as linalool responsible for imparting rose-like aromas. The terpenol molecule is a powerful odorant, even in tiny quantities; however, it is often bound (glycosylated) to a sugar molecule and is therefore odorless. If an appropriate enzyme is available, it can accept the bound terpenol in its active site, break the terpenol–sugar bond, and release the terpenol and sugar molecules separately. The now-freed terpenol molecule is said to be aroma-active, i.e. it can be smelled.
As some enzymes can have unwanted effects, they often need to be deactivated — this is accomplished by blocking their active site, making it unavailable to substrates.
Polyphenoloxidases, or PPOs, are one type of well-known naturally occurring enzymes in musts. PPOs cause phenolic acids to oxidize into brown-colored phenolic compounds. As we know, sulfur dioxide (SO2) is very effective in inhibiting PPOs and preventing phenolic browning.
Laccase enzymes, however, found in must produced from moldy grapes and which also catalyze oxidation of polyphenols into brown-colored pigments, are very difficult to control and their activity can persist throughout the life of the wine. SO2 has little effect on laccases in musts; these can only be inhibited by a thermal treatment at 45 °C (113 °F) or more.
Enzymes fall into one of six classes identified by an EC (Enzyme Commission) number based on the chemical reactions they catalyze, and are easily identified by their usual suffixes ase or yme. Enzyme names usually give a good indication of substrates they act on and the mode of action. PPOs and laccases belong to the class of oxidoreductases, or EC 1, as they catalyze oxidation–reduction reactions.
In winemaking, we will be mainly interested in hydrolases (EC 3), i.e. enzymes that catalyze the hydrolysis of various bonds in molecules, and some lyases (EC 4), i.e. enzymes that cleave various bonds by means other than hydrolysis and oxidation.
Winemaking enzymes
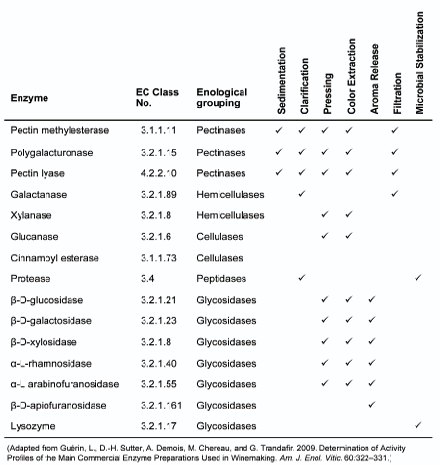
In winemaking, exogenous (enological) enzymes have long been used to improve color and tannin extraction in reds, increase pressing efficiency and yields, boost release of aromas, improve sedimentation (settling), clarification and filtration, and increase microbial stabilization. These enzymes are more commonly grouped and referred according to their enological functions.
The table found at the end of this page lists a selection of common winemaking enzymes and their intended applications along with their EC numbers if you want to get more information; you can search on EC numbers in online enzyme databases such as www.brenda-enzymes.org or www.chem.qmul.ac.uk/iubmb/enzyme.
Pectinases cleave bonds in pectins, those long-chained polysaccharides found in the cell walls of all plant tissues and predominantly in the skins of grapes, making reds at higher risk of developing pectin haze owing to the longer skin contact during maceration and fermentation.
When grapes are crushed, pectins solubilize and form a colloid and bind with grape solids and other macromolecules, and thus interfere with flocculation and cause a haze. As they are more soluble in water than in alcohol, pectins may cause cloudiness well after the wine is bottled. It is more problematic in high-acid wines because pectins become less stable with increasing acidity and cause to depolymerize into smaller compounds. Pectins must therefore be broken down (hydrolyzed) and removed during winemaking using pectinases; grapes alone do not manufacture a sufficient quantity of these enzymes and S. cerevisiae yeast has limited pectinase activity.
Pectinases can also increase yield in the order of up to 5% when used with hemicellulases and cellulases, improve color compound extraction during maceration in red winemaking or rosé winemaking using the saignée method, and improve filterability by reducing filter medium clogging. Lallzyme EX™ and Scottzyme® Color Pro are examples of popular pectinases used as macerating enzymes to act on pectins and enhance color and tannin extraction.
Hemicellulases and cellulases break down hemicelluloses and celluloses, those long polysaccharide chains of glucose and other monosaccharides, such as xylose, mannose, galactose, rhamnose and arabinose, which form part of the supporting structure in wood and plants. As these enzymes tend to become deactivated by ethanol, they are used at the pre-fermentation stage. Hemicelluloses and celluloses are often referred by the broader group name of glucans, and so, the enzymes are also known as glucanases.
Much like pectins, hemicelluloses and celluloses can cause clarification and filtration problems given their high molecular weight — celluloses can consist of 10,000 or more monosaccharides. These enzymes greatly improve these processes in addition to improve pressing and color extraction.
Peptidases, also known as proteases, are proteolitic (i.e. protein-based) enzymes that break down haze-causing proteins by hydrolyzing peptide bonds and improve clarification and filterability. Their activity is limited to pre- and early fermentation where they are also used as a source of yeast nutrients but then become inhibited with increasing levels of ethanol. Given this, peptidases are not used frequently in winemaking particularly that bentonite has proven most effective in dealing with proteins in musts or wines.
Glycosidases are the most important enzymes from an organoleptic perspective as they are responsible for hydrolyzing glycosidic bonds in (non-volatile, odorless) aroma precursor compounds and releasing the bound volatile, aroma-active fraction from their binding fraction — that’s why you can only smell certain aromas after fermentation or enzymatic activity. One of these enzymes, β-D-glucosidase, is also one of the main enzymes produced and secreted by S. cerevisiae yeast.
Glycosidases can also improve pressing yield and color extraction. But note that glycosidases can be inhibited by glucose and therefore are most effective in dry wines, hence why these enzymes need to be added at the end of the alcoholic fermentation. DSM Food Specialties’ Rapidase AR 2000 is an example of a glycosidase used to enhance varietal expression in Muscat varietals, Gewürztraminer and Riesling by releasing aromatic terpenols from their bound fractions.
Because of its mode of action, lysozyme belongs to this group although its enological use is very different from other glycosidases in this group. Lysozyme is a proteolitic enzyme used as a stabilizing agent to safeguard against lactic acid bacteria (LAB) microbial spoilage, particularly after malolactic fermentation (MLF) in reds. Lysozyme activity is most effective in white wines; in reds, the increasing concentration of phenolics during maceration and alcoholic fermentation reduce lysozyme activity as phenols interact with proteins, and so, lysozyme use must be supplemented with SO2. Lysozyme is most active at high pH, when SO2 is least effective, although its inhibitory effect decreases under environmental conditions favorable to the growth of spoilage LAB.
Using enzymes
As there are many enzyme preparations, some of which include two or more types of enzymes, from many vendors, cross-reference the list of ingredients (i.e. what enzymes they contain) with those in the table below to determine their application(s), or consult the manufacturer’s spec sheet, and follow recommended usage, dosage rates, and timing of addition.
Be particularly attentive to environmental conditions including temperature, dosage rate, contact time, SO2 levels, ethanol concentration, presence of fining agents, and phenolic content, as these can greatly affect enzyme performance. Of these, temperature and bentonite are the most critical. High temperatures can denature (and deactivate) proteolitic enzymes while low temperatures can considerably slow down enzymatic activity. Even at low dosage rates, e.g. 3.5 g or one-eighth of an ounce per carboy, bentonite can inhibit enzymes by binding onto their activate sites. And so, enzymes should be used prior to any bentonite treatment. Gelatin and silica gel have a lesser impact on enzyme performance but should also be added following any enzyme use. So be sure to read and follow to the letter the manufacturer’s instructions.
Enzyme preparations are available in dry or liquid forms. Those in dry form need to be dissolved in water, usually in a volume equivalent to 10 times the dry weight, before adding to must or wine. Note that dosage rate is often expressed in g/100 kg of crushed fruit or g/hL of must or wine. 100 kg is 220 lbs and g/hL means grams per hectoliter, where a hectoliter is equivalent to 100 liters or roughly 25 gallons. To figure out a good approximation of dosage rate in grams per 23-L/6-gal carboy, simply divide g//hL by 4; for example, a rate of 20 g/hL would require 5 g per carboy.
Common Winemaking Enzymes and Their Applications