Phenolics & Tannins in White, Sparkling & Rosé Styles

An understanding of what is happening in wine on a chemical basis can be very useful in influencing choices regarding processing options and timing of activities for different wine styles. While we usually don’t think about polyphenolics much when it comes to white wine production, there are definitely some processing choices and stylistic options where polyphenolics can be a concern. In this article we will explore how polyphenolics are extracted into white wine and some options to consider when processing white, sparkling, and rosé wine styles.
Phenolics as a Function of Temperature
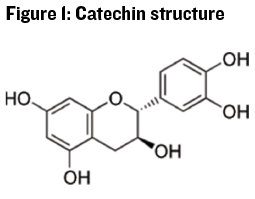
There are different types of polyphenolic compounds in wine and the chemical structure of these compounds impacts their perception in terms of color, bitterness, and astringency. In particular monomeric and polymeric flavonoids, most commonly drawn as catechin subunits (see Figure 1), are important in white wine production in terms of color and bitter flavor.
“tannin” polymers in wine.
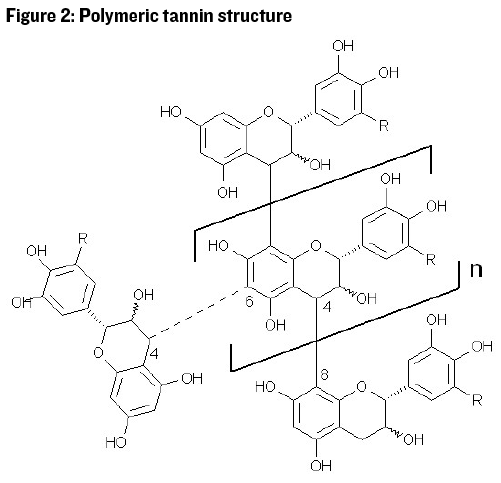
Catechins and similar monomeric phenolic compounds are associated with bitter taste perception. Catechins can exist as monomers (single units) in juice, but most of the catechin is polymerized into larger groups: Two to 16 catechin sub-units in the seeds of grapes, and three to 83 subunits in the skins.1 See Figure 2 for an example of catechin polymerization, where the structure of the polymer may be linear or branched.
Catechin-based compounds (i.e. tannins) have a couple of important impacts in white wine. Two of these important impacts are in taste perception. Tannin compounds may be perceived as bitter tasting; the lingering flavor after drinking black coffee or strong tea without added sugar or cream. Tannin compounds may also be perceived as astringent, creating a feeling of dryness or roughness in the mouth as tannin compounds interact with the proteins in saliva.
Catechin monomers and smaller catechin polymers are generally described as having a bitter taste, but little impact on the feeling of astringency. Larger catechin-based polymers of four or more units have the opposite effect, with limited bitterness but very impactful in perception of astringency.
Catechin-based polymers are also indicative of the browning potential for white wines. Catechin and epicatechin (a stereoisomer of catechin also present in grapes) participate in coupling reactions with other phenolic substances (hydroxycinnamic acids) to make compounds that appear yellow to brown colored. Research has shown that higher levels of catechin-based compounds increase the browning potential of white juices and wines.
As discussed previously, catechin monomers and short polymers are found more in seeds, whereas longer chains are found in skin tissue. However, catechin monomers and small polymers may be found in the skin of unripe fruit. Catechin monomers are also more easily extracted from the seeds of unripe grapes.2 Thus, catechin size, and therefore sensory impact, is influenced by berry ripeness as well as by grape variety. Hybrid grapes may have more catechin than vinifera grapes, and are often grown in climates where berry ripeness is more difficult to achieve. Therefore hybrid fruit may be especially prone to browning reactions and bitter taste perception.
With any given amount of polyphenolics in grape berries, controlling the white wine processing decisions with respect to tannin management will control the color/browning potential and the bitterness and astringency found in the finished wine. Because polyphenolic molecules are found in skins and seeds, options on the crush pad (the temperature and amount of time that juice is allowed to interact with skin and seed solids) will control the type and amount of polyphenolics extracted into the juice. So for most white wines, and especially for sparkling wine production, limiting phenolic pickup is important both to limit browning potential and also to limit bitterness and astringency. During the pressing process, one way to limit phenolic pickup is to whole-cluster press. By whole cluster pressing you eliminate phenolic extraction during the destemming and crushing step, and also during the time when the press is being filled. However, without an automated membrane press the whole cluster press method is either going to produce very low juice yield or be very time-consuming (multiple sequences of breaking apart the cake and re-pressing), so most home wine producers will probably have to destem and crush to use their basket press.
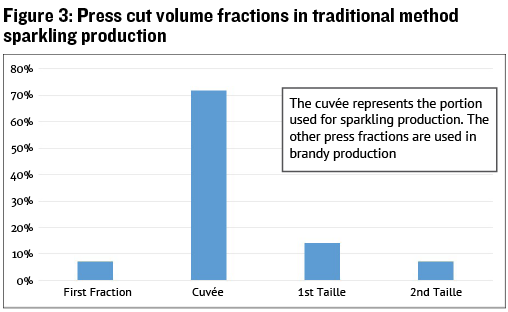
No matter what type of press is used, press cuts can also play an important role in limiting phenolic pickup. I wrote an entire article on press cuts titled “Making the Cut” in the August-September 2018 issue, but to summarize for white wine production: Phenolic extraction increases dramatically in approximately the last 25% of the press volume, so splitting this portion of the press run from the main juice volume allows you to either use this portion for another purpose, or to treat the hard-press juice with fining agents before blending back with the main batch. An example of this may be seen with the common scheme for traditional sparkling wine production, illustrated in Figure 3.
Besides the type of press and use of press cuts, there is another opportunity for limiting phenolic pickup: Temperature. Lower temperatures will yield lower phenolic extraction, so harvesting and pressing before midday temperatures rise, or utilizing refrigeration of fruit prior to pressing, will limit the extraction of phenolic compounds into the juice. Limiting phenolic extraction, along with a desire for lower temperatures during cold settling (after pressing) to prevent premature fermentation, is the reason why many commercial winemakers will not continue to harvest after mid-morning.
One aspect of white pressing we haven’t discussed yet is anthocyanins — the polyphenolics responsible for red and purple colors in grapes. Obviously most white grapes are deficient in anthocyanins, but there are several grape varieties used to make white wines (e.g. Pinot Gris and Gewürztraminer) that have markedly pink berries and can produce colored juices. Just as with phenolic compounds in general, limiting temperatures and skin contact time will limit the amount of color in the pressed juice of these varieties. Some color is clearly warranted for rosé wine production, and most winemakers will soak rosé juice on the skins for 4–48 hours before pressing off the juice. Anthocyanins are relatively quick to extract into juice, with the maximum color often extracted in the first one to three days of maceration for red wines. So for rosés, cold temperatures can be important during processing to limit the extraction of bitter and astringent phenolic compounds while the winemaker waits for the optimum color expression. Keep in mind that some color will be lost during fermentation, fining, and filtration, so it’s best to press when the juice is just slightly darker than the color depth desired.
There are several instances where extraction of phenolic compounds is inevitable due to other processing limitations or stylistic choices. Use of a basket press, mentioned earlier, is an example where crushing and destemming is necessary to obtain adequate juice yields. Rosé production is also going to result in some phenolic extraction. Another example is the use of cold soaking for some white varieties in order to maximize aromas. In the Finger Lakes region of New York, where I live, it is common to allow varieties such as Riesling to soak on their skins overnight before pressing. Research has shown that there are significant levels of aroma molecules bound to skin tissue in grape berries, and some of these aroma molecules are extracted into the juice during skin contact. Phenolic molecules are generally too small to be filtered out of wine unless tannin polymers become so large they precipitate and form a solid. So, what do winemakers do when faced with color precursors, bitter phenolic molecules, or astringent tannin in whites? They turn to fining agents.
The role of fining
The concept of wine fining is the addition of a compound that attracts or binds to a target compound that is then removed from the wine. The fining agent must be a compound with limited solubility so that it can be easily removed by racking the clear wine away from the sedimented fining agent. The fining agent must also attract the targeted compound and will hopefully have limited attraction to other compounds. Phenolic compounds have many hydroxide (OH) groups (see Figure 2) that tend to carry a slight negative charge in solution. So, to fine out phenolic compounds we need to add a fining agent with a slight positive charge that will attract and loosely bind the phenolic compound.
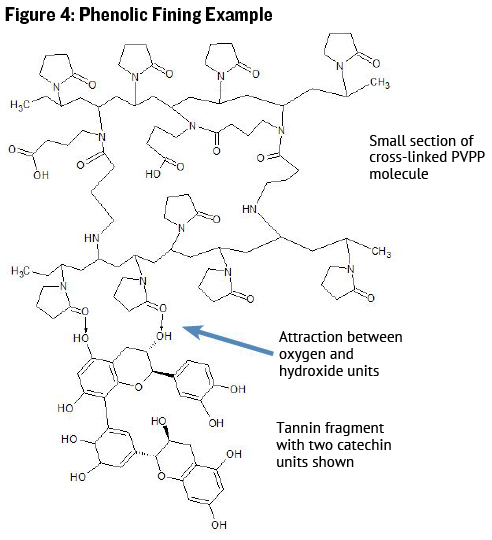
One of these types of compounds approved for use in wine production is a plastic polymer called polyvinylpolypyrrolidone (PVPP). PVPP is a crosslinked polymer that has many pyrrolidone units that contain a ring-connected oxygen atom that carries a slight positive charge in solution. Thus the slight positive charges on the PVPP molecule attract the slight negative charges on the phenolic molecule (see Figure 4), and since the PVPP is insoluble (doesn’t dissolve in juice or wine), the wine may be racked or filtered leaving the PVPP and phenolic molecules behind.
Another group of fining agents with a slight positive charge in solution are proteins. The most common protein-based fining agents for white juices and wines are gelatin, isinglass, and casein. Of these, gelatin and casein are often utilized for fining browning or pinking precursors as well as bitter and astringent phenolic compounds. Between gelatin and casein, gelatin is probably the easiest to work with and is a common tool in commercial white wine production. Comparing PVPP to gelatin, PVPP is thought to attract browning precursors and smaller phenolic compounds that contribute to bitter taste, while gelatin is thought to attract larger phenolic molecules that contribute to astringency. However, there are several different types of gelatin-based fining agents, with different size ranges, that are thought to attract a slightly different range of phenolic compounds. It is necessary to read the manufacturer recommendations or consult with the supplier to obtain the best product for a given application. Follow the manufacturer’s directions for dosing rates at the juice stage. For finished wines, once a product or two has been selected as a candidate for fining it is important to perform a benchtop trial to decide the optimum agent and dosage rate based on aroma and taste.
In summary, we’ve discussed some methods that can limit phenolic pickup in white juice as well as some fining agents that can decrease the level of phenolic molecules in both juices and wines. In the next part of this phenolic series we’ll explore the complexity of phenolic molecules in red wine production.
References
1 Johnson, C. (2016). Tannin – Key Factors in Red Wine Taste and Mouthfeel. Retrieved from UC Davis Waterhouse Lab – What’s in Wine?: https://waterhouse.ucdavis.edu/whats-in-wine/tannin
2 ETS Laboratories. (2020). Phenolic Composition – Profiling phenolic compounds in red wine. Retrieved from ETS Laboratories: https://www.etslabs.com/library/34






