Q
Here’s one problem you may have been asked about before. My finished wine has an aroma and taste of rubber and burnt toast, a bit like burnt rubber. Since I will be making more wine this year, I sure would like to know what I’m doing wrong. Would you let me know?
Richard Larocque
L’Orignal, Ontario
A
I can think of two possible causes of these flavors and smells. The first is over-oaking of your wine with highly toasted chips. If you are using chips, run bench trials to see what level of oakiness you like. Take small samples of your wine and add oak chips at varying rates. Taste the wine and see what level you best enjoy. If this is too involved, play it safe and go with a low rate like 1 g/L.
The second possible cause is hydrogen disulfide or mercaptans. There are numerous potential pathways for these stinky compounds to get into your wine. Sulfur spraying in the vineyard too close to the harvest date is one possibility. Yeast spit out elemental sulfur in the form of hydrogen sulfide. Other causes might include insufficient nitrogen levels during fermentation, high fermentation temperatures, low fermentation temperatures, a too-rapid fermentation or anything that will stress your yeast out. Most often, hydrogen sulfide smells like rotten eggs and mercaptan smells kind of oniony, but I’ve seen them develop into burnt rubber aromas in finished wines.
Two possible solutions to this problem are aeration or adding copper sulfate. While the wine is still fermenting, you can try aerating it, but don’t aerate after fermentation is complete. Alternately, you can add copper sulfate, which binds to hydrogen sulfide. Copper sulfide should only be used at concentrations lower than 0.5 mg/L (ppm). (I discussed this in more detail in the Winter 2001 Winemaker.)
Sometimes hydrogen sulfide and especially mercaptans can’t be removed at all and even professional folks have to live with the stinky results of naughty yeast. Never fun, but a reality of the business.
Q
I seem to be having a recurring problem of excess CO2 (bubbles) in my wines. The wines are bubbly long after fermentation is finished. They have all been racked at least once and are stored in glass carboys. What do you suggest I do to get the wines settled down prior to bottling?
Laurie Hammack
via email
A
Unless you artificially carbonate your still wines by kegging them, there is only one possible source of carbon dioxide in wines — microbes, specifically little bacteria and yeastie beasties that love to inhabit wine even after all of the fermentable sugar has been used up.
If you’ve measured your wine for dryness by using a hydrometer — and it sounds like you have — you’re most likely experiencing the joys of a prolonged malolactic fermentation. Malolactic fermentations occur when certain strains of bacteria eat the malic acid naturally present in grapes and spit it out as lactic acid and carbon dioxide gas. Some strains are better for wine quality than others, but many common malolactic bacteria can be found just floating in the air around us. There are some less common spoilage microorganisms that produce carbon dioxide, but malolactic bacteria is likely your number-one culprit.
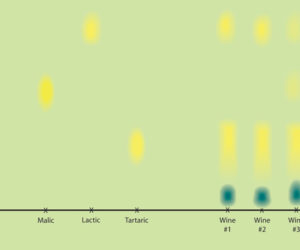
In order to get the carbon dioxide to come out, I suggest you take the patient approach — wait. Eventually the malolactic bacteria will consume all of the malic acid and will no longer produce any more carbon dioxide. Don’t rack the wine again during your waiting period, because the lees in the carboys can actually help the bacteria finish their job by providing them with micronutrients and other goodies they need to survive. Racking will also aerate the wine and inhibit the malolactic bacteria, as they are anaerobic. Keep your wine out of the cold — keep the temperature above 60 °F (16 °C) — so that the malolactic bacteria aren’t killed off and can keep working. In the meantime, try to keep your containers topped up in order to stave off any micro-aerophilic spoilage organisms (like the vinegar-producing Acetobacter). You can test for the completeness of your malolactic fermentation by using a paper chromatography kit, available through most home winemaking supply stores or mail-order catalogs.
Once malolactic fermentation is complete, rack the wine off the lees, add sulfur dioxide if desired and bottle as usual. One of the reasons that winemakers actively encourage malolactic fermentation is that it lessens the need for pre-bottling filtration. If there is no more malic acid left for the malolactic bacteria to eat, it’s not a problem if some of them get through the filter and into the bottle . . . unless, of course, you want a little fizz in your wine later on down the road!