The Power of Tannins
In their authoritative Handbook of Enology, Pascal Ribéreau-Gayon — one of the most influential enologists of the 20th century — and his collaborators wrote: “Tannins are among the most important constituents of wine, influencing not only taste and color, but also its capacity for clarification, stabilization, and aging.”

Despite this central role, tannins are often discussed by home winemakers primarily in terms of harshness or bitterness. In reality, tannins comprise a diverse family of compounds whose chemical behavior during crushing, fermentation, fining, and aging profoundly affects wine structure and mouthfeel, color in the case of reds, and shelf life. When properly managed, tannins make wine taste richer and fuller-bodied while also improving color stability, clarity, and longevity. When excessive or poorly extracted, however, tannins can make a wine taste bitter, excessively astringent, and potentially unapproachable for years until they have time to “soften.”
Understanding where tannins come from, how they differ, and how they can be managed — specifically, how to extract desirable tannins from grape skins while avoiding harsher tannins from seeds and stems, and when to supplement juice or wine with exogenous tannins — allows winemakers not only to avoid faults, but also to shape the style and aging potential of their wines.
This article explores the different types of tannins, their sources, how they function and interact with other wine compounds, and their specific applications in winemaking. Given their more significant role in reds, the focus here is on red winemaking.
Types of Tannins
Tannins are found in grape skins, seeds, and stems, as well as in certain woods and nuts, notably oak and chestnut. Tannins do not register as a flavor in the traditional sense, but as a tactile sensation. When tannins bind with saliva proteins, lubrication in the mouth is reduced, creating sensations described as drying, rough, or puckering. This is why tannins can feel harsher in young wines and smoother as wines age and tannins polymerize into larger, less reactive molecules. High acidity reinforces both bitterness and astringency, which explains why tannic red wines are typically produced with lower acidity.
Chemically, tannins belong to a class of compounds known as phenols. Because they tend to form increasingly large polymers with other phenols, they are commonly referred to as polyphenols. In winemaking, tannins are still classified as either condensed or hydrolyzable — a distinction adopted long before modern analytical techniques clarified their detailed molecular structures.
Condensed tannins (proanthocyanins)
Condensed tannins — more precisely called proanthocyanins because they release anthocyanins (color pigment molecules) when heated in an acidic medium — are found in grape skins, seeds, and stems. These tannins impart a firm, dry mouthfeel and play a major role in building wine structure. They also possess strong antioxidant capacity and excellent color-stabilizing power, as we’ll discuss later.
Among grape-derived tannins, skin tannins are the most desirable. They tend to be riper, less bitter, and less astringent than seed or stem tannins, and winemakers aim to extract them as fully as possible when producing full-bodied red wines. Seed tannins, by contrast, are more bitter and more astringent, and winemakers generally try to limit their extraction during fermentation. Stem tannins are the harshest of all, especially when stems are green rather than fully lignified. Stems also contain relatively high levels of potassium (K+), which can affect pH, color stability, and tartrate stability. For these reasons, most winemakers avoid stem contact by destemming grapes prior to fermentation.
Condensed tannins can also be derived from certain exotic woods, particularly quebracho (Schinopsis spp.), a South American hardwood that is exceptionally rich in extractable proanthocyanins.
Hydrolyzable tannins
Hydrolyzable tannins differ fundamentally in structure from condensed tannins and are so named because they hydrolyze into ellagic or gallic acid in acidic conditions. Ellagic acid tannins, or ellagitannins, are found primarily in oak (Quercus spp.) and in even higher concentrations in chestnut (Castanea spp.). Gallic acid tannins, or gallotannins, are found in gallnuts, cherry wood (Prunus spp.), and tara, a leguminous tree native to Peru and the Andes.
Although both ellagitannins and gallotannins are classified as hydrolyzable tannins, their chemical differences from proanthocyanins are significant. Both exhibit greater antioxidant power than grape-derived tannins, which makes them particularly useful for protecting juice and wine in white and rosé winemaking. Proanthocyanins and ellagitannins contribute strongly to color stabilization and tend to produce a more harmonious astringency, whereas gallotannins have limited color-stabilizing ability and are generally perceived as more bitter.

Functual Classification and Applications
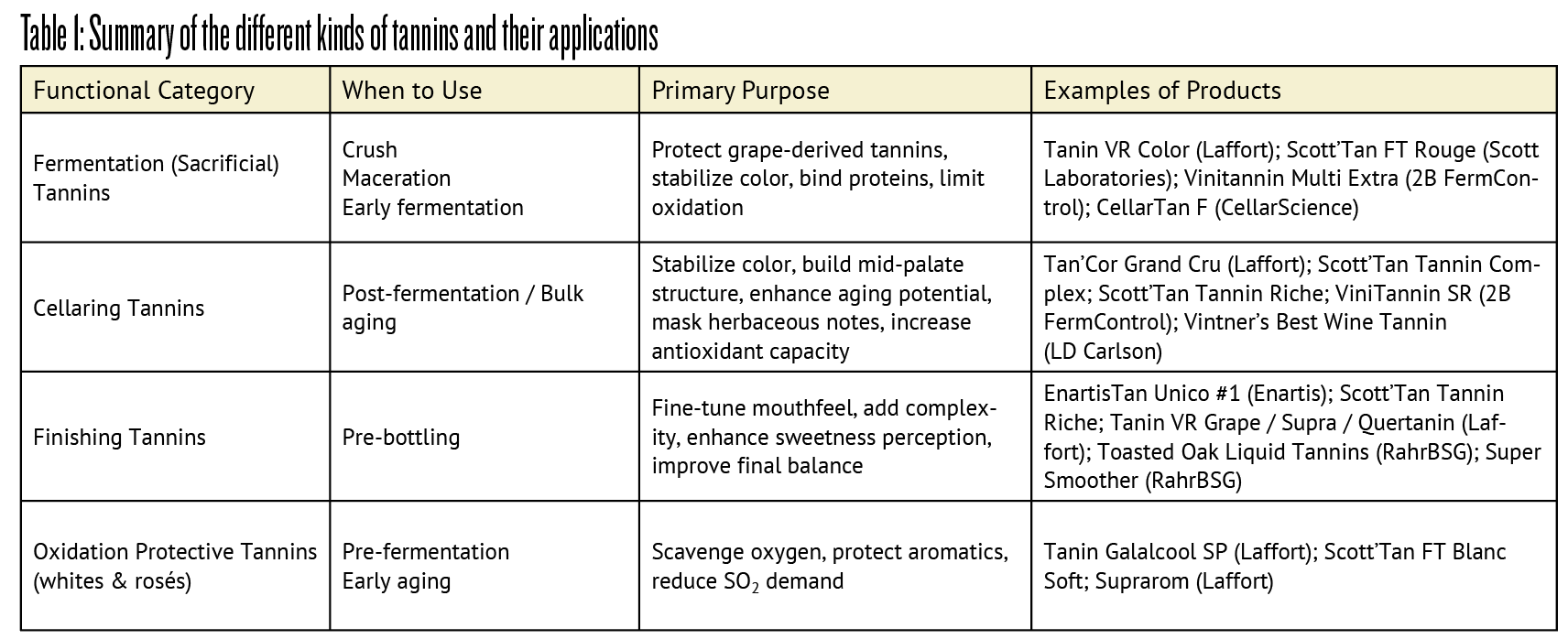
In practice, tannins are often classified by function rather than origin. They may be described as fermentation (or sacrificial) tannins, cellaring tannins, or finishing tannins, depending on when and why they are used. Tannins can be added pre-fermentation (at crush or during maceration), during fermentation, during bulk aging, or shortly before bottling to fine-tune the final wine. Table 1 summarizes the main tannin types, their applications, and representative commercial products.
Note, although often used for similar purposes in adjusting bitterness and mouthfeel, products such as Opti-Red, Noblesse, and Oenolees are yeast-derivative products, not tannins. The use of inactivated dry yeast (IDY) for body and mouthfeel adjustment is discussed in the article “Shaping Body and Mouthfeel in Wine.”
The Chemistry of Tannins
The chemistry of tannins is complex and, in many ways, fascinating — particularly as it pertains to their interactions with oxygen, sulfur dioxide (SO2), and anthocyanins (color pigment molecules).
In the presence of oxygen and naturally occurring oxidation catalysts — namely, iron and copper — tannins gradually oxidize, shifting in color from orange to brown as they transform into compounds known as quinones. Remarkably, tannins can regenerate from their quinone forms in the presence of SO2. This reversible behavior underlies their effectiveness as antioxidants and explains their critical role in protecting wine from oxidative damage during aging.
Tannins also enhance color stability in red wine by interacting with anthocyanins to form increasingly large and stable tannin — anthocyanin-tannin polymers. Anthocyanins are highly reactive and unstable on their own and are strongly influenced by pH. Without sufficient tannins, anthocyanins can react undesirably or precipitate, leading to diminished or unstable color.
A richly colored red wine that contains very little tannins may deteriorate quickly. This is particularly evident in nouveau-style wines, such as Beaujolais Nouveau, which are produced using carbonic maceration where whole grape clusters are fermented without crushing (to maximize fruit aroma extraction while limiting tannin extraction). As a result, these wines are intended for early consumption as they have no aging potential.
Conversely, a wine with abundant tannins but little anthocyanins (poor color) will be unbalanced, tasting excessively bitter and highly astringent. Achieving the right balance between tannins and anthocyanins is therefore one of the central challenges — and arts — of red winemaking.
Grape varieties differ widely in their natural levels of anthocyanins, tannins, and tannin-binding proteins. Richly colored but low-tannin varieties, as well as high-protein varieties (including many North American and hybrid grapes), often benefit from tannin additions to achieve balance. Wineries equipped with analytical tools can fine-tune tannin and anthocyanin levels to specific ratios; a tannin-to-anthocyanin ratio of approximately 4:1 is often considered ideal.
pH plays a critical role as well. The recommended pH range for red wines is 3.3–3.6. Beyond increasing susceptibility to microbial spoilage, high pH alters color by shifting anthocyanins toward their colorless forms. For perspective, at pH 3.3 a typical red wine may contain less than 20% of anthocyanins in their red-colored form, whereas at pH 4.0 this can drop below 5%. Maximizing color extraction and ensuring an adequate supply of tannins therefore becomes increasingly important at higher pH.
In addition to their effects on color and oxidation, tannins act as natural fining agents by binding and removing proteins that can cause haze. They also exhibit antimicrobial activity, allowing for lower free SO2 levels in red wines. For example, if a typical target is 0.5 mg/L molecular SO2, full-bodied, tannic reds may remain protected at 0.2–0.3 mg/L. However, when barrel aging, a minimum of 0.5–0.6 mg/L molecular SO2 should be maintained, as acetic acid bacteria can thrive in barrel headspace at lower levels, leading to elevated volatile acidity.
Fermentation (Sacrificial) Tannins
During maceration and fermentation, grape skin tannins are extracted into the juice and wine, where they are exposed to oxygen and bind readily with naturally occurring proteins. Both oxidation and protein binding reduce the pool of desirable tannins, potentially compromising color stability, structure, and aging potential.
To mitigate this loss, winemakers can add fermentation or sacrificial tannins at crush or during maceration. These tannins preferentially oxidize and bind with proteins, thereby preserving grape-derived tannins for color stabilization and structural development.
Fermentation tannins are commonly added using commercial grape tannin products or powdered oak or gallnut preparations, which release ellagitannins or gallotannins, respectively.
Cellaring Tannins
After fermentation, red wines that are not barrel aged often benefit from tannin supplementation during bulk aging. Cellaring tannins help maintain color stability, reinforce mid-palate structure, and enhance antioxidant protection.
They can also be useful in masking herbaceous or green bell pepper aromas caused by methoxypyrazines (pyrazines), which are found in Cabernet Sauvignon, Cabernet Franc, Merlot, Carménère, Malbec, and related varieties. Pyrazines decrease with grape ripening, but when present at elevated levels they impart vegetal aromas such as green pepper, cut grass, and asparagus. These aromas can mask fruit character and are notoriously difficult to remove once established.

Finishing Tannins
Finishing tannins are used shortly before bottling to fine-tune mouthfeel, body, and overall complexity. Depending on the source, they may contribute subtle oak-derived notes such as coconut or vanillin, or enhance the perception of sweetness without adding sugar.
Sourcing and Adding Tannins
All tannins discussed here are available as commercial enological preparations. For home winemakers, sourcing can be challenging, as many products are sold in winery-scale formats or repackaged with limited usage information. Sharing larger quantities within a winemaking club and consulting retailer websites for detailed technical sheets can be helpful.
Because tannins vary widely in sensory impact and are strongly influenced by alcohol and acidity, bench trials are critical. Over-addition is easy and difficult to reverse. The following guiding principles, adapted from Émile Peynaud’s The Taste of Wine (Macdonald & Co, 1987) are useful when seeking balance:
Acidity, bitterness, and astringency reinforce one another. High-acid wines intensify tannin bitterness and astringency; therefore, highly tannic wines should have lower acidity, and high-acid wines should have lower tannin levels.
High acidity or high bitterness is best balanced by higher alcohol. Avoid combining low acidity and low tannins with high alcohol, or high acidity and high tannins with low alcohol.
Sweetness and acidity counteract one another. This principle is particularly useful in softening high-acid whites and rosés by retaining a small amount of residual sugar.
As with all tannin additions, bench trials are essential. Determine the optimal dosage, allow at least a couple of weeks for integration, reassess the wine, and make further adjustments, if necessary, before bottling.
Conduct bench trials using 100-mL samples at varying dosages and compare them against a control. Once the preferred level is identified, treat the entire batch, then re-evaluate after two to four weeks. Follow product-specific instructions carefully, as some tannins must be dissolved prior to addition. Expect tannin additions to produce sediment, and so, plan to rack before bottling.
To address pyrazines, add untoasted or lightly toasted oak (or a blend) at a rate of 2–4 g/L (0.25–0.5 oz./gallon). Assess aroma weekly and remove the oak or rack the wine once the vegetal notes are sufficiently masked. Finishing tannins can then be added to enhance complexity, following bench trials.
Tannin additions are powerful and difficult to reverse. Always conduct bench trials using multiple dosages alongside a control sample. Allow sufficient integration time before making final decisions. What tastes assertive today may integrate beautifully with time.
Tannin Management for Advanced Winemakers
Winemakers with access to a spectrophotometer can assess tannin levels quantitatively. Tannin concentration is commonly estimated using the Total Phenol Index (TPI), determined either by the Folin-Ciocalteu assay or by measuring absorbance at a wavelength of 280 nm.
Typical values are below 20 for whites and rosés, 20–30 for light reds, and 30–50 for well-structured reds. Wines exceeding a TPI of 50 are generally considered very tannic and potentially overly astringent. Astringency can be evaluated by measuring absorbance at 230 nm.
For red wines, measurements at 230 nm and 280 nm should be performed on samples diluted 1:10 and analyzed using 1-mm UV or IR-type cuvettes.
There are tools and models (e.g., Skogerson–Boulton Model Assay) to analyze spectral data to quantify more precisely tannins and anthocyanins and evaluate tannin-to-anthocyanin ratios.
Put Your Tannin Knowledge to Use
Now that you know the different types of tannins, it’s time to put that knowledge into practice. Check out these techniques on using tannins in your winemaking.
Using Tannins: Purposes, sources, and use in winemaking
Balancing Wine’s Structure: Techniques to add and remove tannins






